- Home
- About Us
- Work
- Journal
- Contact
- Bairi piya english translation
- Portrait professional 11 manual
- Adobe reader 8
- Viralukketha veekkam movie
- Up board hindi books
- Lumerical fdtd
- Isuzu parts tucson
- Fire emblem rom hacks how to make
- Oxygen in zno xps peak
- Chord pickout software
- Wonder woman 2017 dual audio
- Frozen synapse soundtrack reddit
- Home
- About Us
- Work
- Journal
- Contact
- Bairi piya english translation
- Portrait professional 11 manual
- Adobe reader 8
- Viralukketha veekkam movie
- Up board hindi books
- Lumerical fdtd
- Isuzu parts tucson
- Fire emblem rom hacks how to make
- Oxygen in zno xps peak
- Chord pickout software
- Wonder woman 2017 dual audio
- Frozen synapse soundtrack reddit

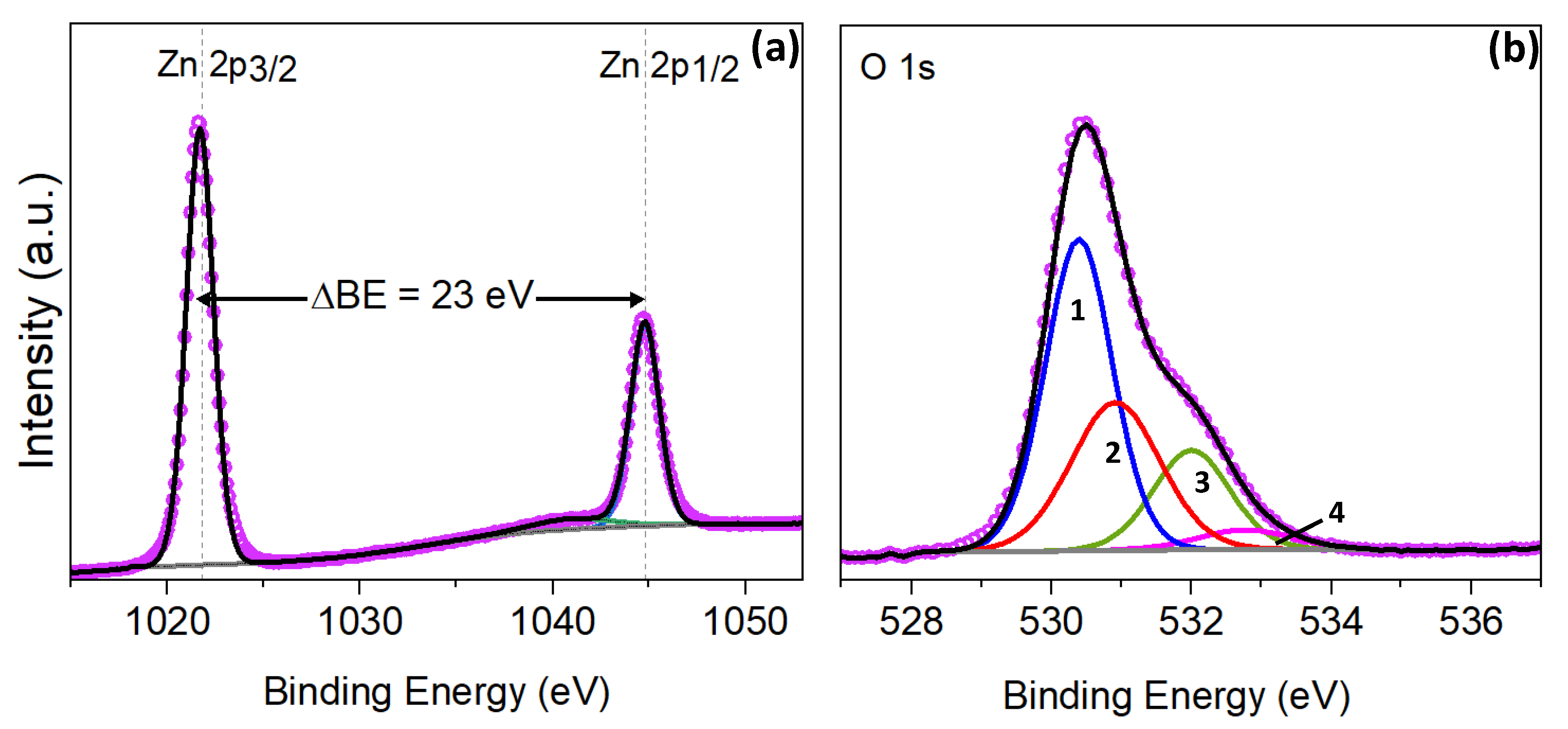
A number of different methods are currently used to fabricate AZO thin films, the most common being magnetron sputtering, pulsed laser deposition, chemical vapor deposition, chemical spray deposition, pulsed laser deposition (PLD), and atomic layer deposition (ALD). IntroductionĪluminum-doped zinc-oxide (AZO) films have been intensively investigated in recent years because their electrical properties make them suitable for a wide range of applications. The area ratio of XPS peaks (A/B) was clearly related to the stoichiometry of AZO films that is, the higher value of A/B showed the higher stoichiometric properties. X-ray photoelectron spectra (XPS) of the O1s were decomposed into metal oxide component (peak A) and the adsorbed molecular oxygen on thin films (peak B).

The X-ray diffraction (XRD) patterns show that the (002)/(103) peak-intensity ratio decreased as the gas flow rate increased, which was related to the increase of AZO thin film disorder. The charge-carrier concentrations in the films decreased from 1.69 × 10 21 to 6.16 × 10 17 cm −3 with increased gas flow rate from 7 to 21 sccm.
#Oxygen in zno xps peak tv
Zinc oxide is used in plastics, cosmetics, and wallpaper, while zinc sulfide is employed in luminescent dials, TV screens, and fluorescent lights.Al-doped zinc-oxide (AZO) thin films were prepared by RF magnetron sputtering at different oxygen partial pressures and substrate temperatures. It is also used to form numerous metal alloys such as brass, bronze, and aluminum solder. Zinc’s principle use is to galvanize iron and steel, preventing corrosion. The growth of plants and animals, and altered biological concentrations of zinc can lead to severe health problems.
This bluish-white metal is brittle at ambient temperatures but becomes malleable between 110 ☌ – 150 ☌. Marggraf is credited for the first isolation of zinc from the charcoal reduction of calamine in 1746. Zinc alloys have been used for centuries, although A.

